Hundreds of lives transformed by SynCardia’s artificial heart
SynCardia, winner of the 2014 New Economy Award for Best Medical Device Company, explains how its temporary Total Artificial Heart bridges the gap to a donor heart transplant
The SynCardia Total Artificial Heart is the only medical device approved in the EU, Canada and the US that eliminates the source of end-stage biventricular heart failure
Cardiovascular disease, which includes heart failure, is the leading cause of death in the civilised world – more than all cancers combined. For years, heart transplants were used to save patients dying from end-stage heart failure in which the heart no longer pumps enough blood to sustain the body.
1,347
Longest (in days) a patient has used the Total Artificial Heart
3,400
Patients in the EU waiting for a donor heart in 2012
2,004
Heart transplants conducted in the EU in 2012
The need for donor hearts is growing as heart disease continues to claim an ever-greater percentage of the world’s population. However, the number of donor hearts available for transplant has been flat in some countries and is declining in others.
Among EU countries, 3,400 patients were on waiting lists for a donor heart in 2012. According to the European Commission’s Department of Health and Consumers, only 2,004 transplants were conducted that year. According to the US Department of Health and Human Services, about 3,800 people wait for a donor heart transplant on any given day, while the supply of approximately 2,300 donor hearts annually has been flat in the US for more than 20 years.
Total Artificial Heart: a life-saving bridge
The SynCardia temporary Total Artificial Heart is the only FDA, Health Canada and CE approved total artificial heart in the world. It has been implanted more than 1,350 times and has provided more than 400 years of support for patients in Europe, Canada and the US.
According to data from the 10-year pivotal clinical study which led to FDA approval, published in the August 2004 New England Journal of Medicine, 79 percent of patients who received the Total Artificial Heart were bridged to transplant. This is the highest bridge to transplant rate for any approved heart device in the world.
Like a heart transplant, the SynCardia Total Artificial Heart replaces both failing heart ventricles and the four heart valves. It is able to provide blood flow of up to 9.5 litres per minute through each ventricle – more blood than an average human heart can pump. The SynCardia Heart is the only approved medical device that eliminates the source of end-stage biventricular heartfailure. Soon after implant surgery, doctors and family members often see patients turn from a sickly grey to a healthier pink as blood flow is restored to their body and vital organs.
Returning freedom following Cardiovascular Disease
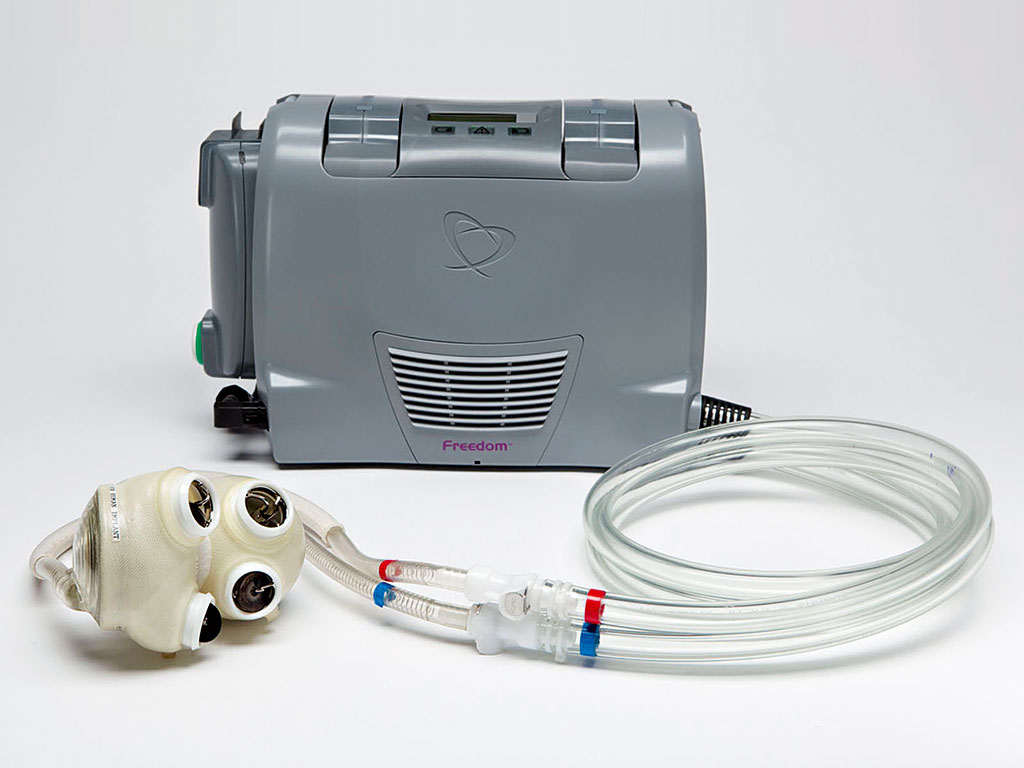
Because of its elegant design, the Total Artificial Heart doesn’t require sensors, motors or electronics of any type inside the body. All of these “wear components” are located safely outside the body in the hospital driver and the Freedom portable driver. There is never a need to re-operate to repair or replace faulty electronics.
As soon as a patient becomes clinically stable, they can be switched to the Freedom portable driver. The Freedom can be worn in a backpack, carried in a shoulder bag or pulled on a rolling cart. Patients with the Freedom portable driver can be discharged from hospital to live at home and in their communities while they wait for a matching donor heart.
Freedom driver patients exercise, eat at home, socialise, recreate and even go back to work. All these activities help the patient get stronger and healthier, which helps lead to better outcomes when the patient receives a matching donor heart transplant. Hospital discharge can also save patients, hospitals and insurance companies thousands of dollars through eliminating most in-hospital costs for this portion of patient care.
The Freedom portable driver empowers SynCardia Total Artificial Heart patients to get on with living their lives while they wait for their heart transplants: Christopher Larsen boxes to stay in shape; Randy Shepherd wore the Freedom driver in his backpack while he walked a 4.2-mile course in an annual running/walking event that honours a fallen US soldier; at 16 years old, Nalexia Henderson became the youngest SynCardia Total Artificial Heart patient to be discharged from the hospital on the Freedom portable driver; Pietro Zorzetto, the man who has lived the longest (1,374 days) on the SynCardia Total Artificial Heart, spent all but two months of that time discharged from hospital.
More than 175 patients worldwide have used the Freedom driver, totalling more than 100 patient years of support and allowing practically unlimited mobility. SynCardia Systems is seeking approval from the FDA for a study into using the SynCardia Total Artificial Heart as a permanent heart replacement, known as destination therapy. The company has also applied for FDA approval of a study into using a smaller 50cc version of the Total Artificial Heart. The smaller heart is designed to fit all women, smaller men and many adolescents.
For further information tel: +1-520-955-0660, or visit syncardia.com













